AG Nicke "Ionenkanal-Rezeptoren"
Leitung: Prof. Dr. nat. phil. Annette Nicke
Postdoc: Dr. Béla Zimmer
Doktoranden: Stefan Fuchs, Jonas Seidler
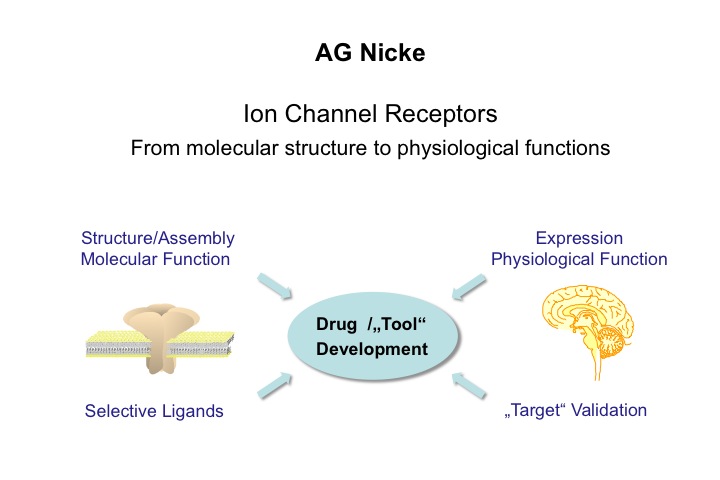
Research interests
- Assembly of membrane proteins (in particular receptors and transporters)
- Structure and molecular function of neurotransmitter-gated ion channels
- Structure-activity relations of peptide toxins
- Physiological and pathophysiological functions of P2X receptors
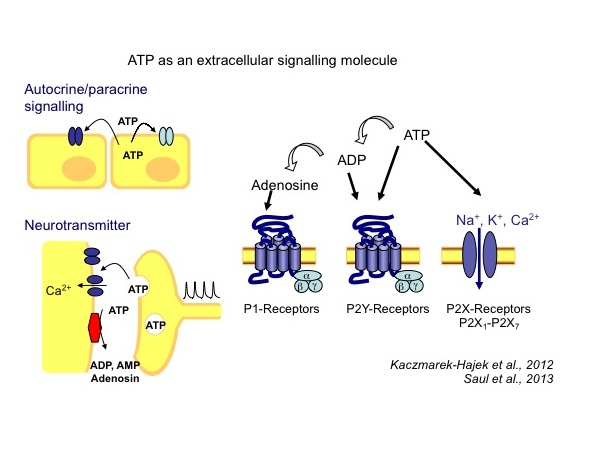
Adenosine 5’-triphosphate (ATP) is an essential molecule for all life forms. Research of the past forty years has shown that ATP is not only a principal energy source and component of nucleic acids inside the cell, but also plays a crucial role in intercellular communication. This possibly oldest transmitter is involved in both fast and slow communication between cells by activating ionotropic P2X (ligand-gated ion channel receptors) or metabotropic P2Y (G-protein coupled receptors) receptor families. Following the cloning of the seven P2X isoforms, it has been shown that P2X receptors represent a novel class of ligand-gated ion channels with unique molecular properties. In particular, the molecular mechanisms underlying the formation of large plasma membrane pores and cell morphology changes upon P2X7 receptor activation remain enigmatic.
P2X receptors are present in virtually all mammalian tissues and mediate a large variety of responses ranging from fast neurotransmission to proliferation and cell death, to only name a few. Their importance as novel drug targets (e.g. for the treatment of pain and inflammatory processes) is increasingly becoming apparent.
Current projects
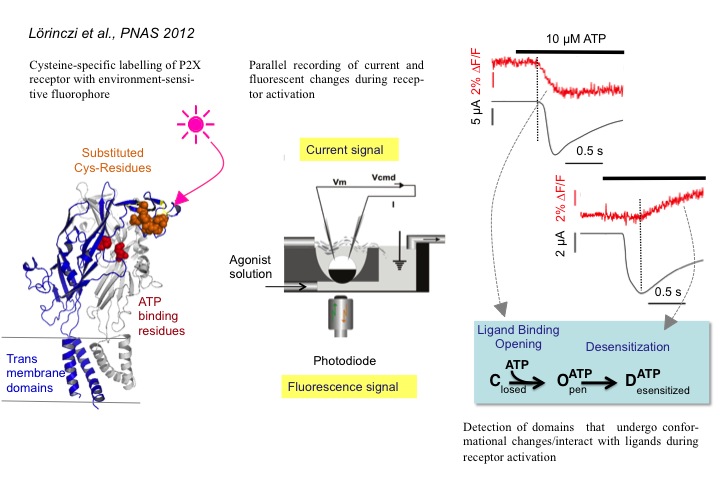
1) Molecular structure and function of P2X receptors
P2X receptors (P2XRs) are ATP-activated, Ca2+-permeable cation channels that are assembled as homo- or heterotrimers (Nicke et al., EMBO J., 1998). Using site-directed mutagenesis and disulfide cross-linking experiments in combination with two-electrode voltage-clamp analysis, we could localize the ATP binding site at the interface of neighboring P2X subunits (Marquez-Klaka et al., J. Neurosci., 2007). To analyze conformational changes that are associated with the activation and desensitization, we now apply voltage clamp fluorometry (Lörinczi et al., PNAS, 2012).
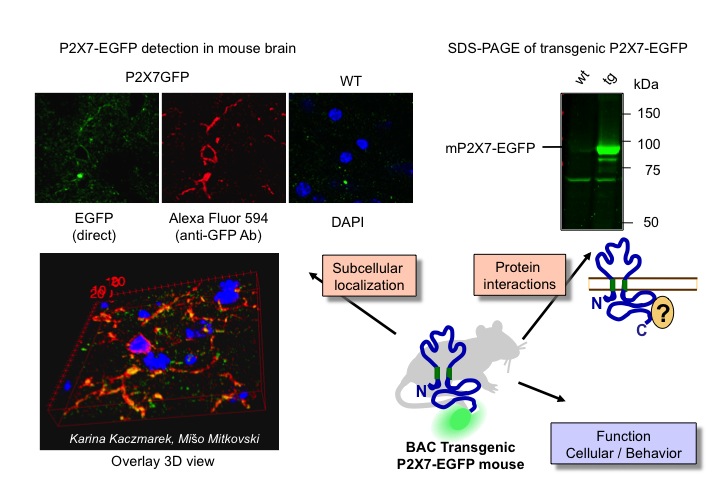
2) Generation of BAC transgenic mice to investigate the expression patterns, protein interactions and physiological functions of P2X receptors
In this project, we have generated bacterial artificial chromosome (BAC) transgenic mice that over-express the P2X7 receptor subtype and allow visualization of its expression pattern in the whole animal as well as investigation of its specific localizations and protein interactions in different cell types. In the long term, we plan to study physiological consequences of altered receptor function upon expression of functionally modified P2X receptor variants.
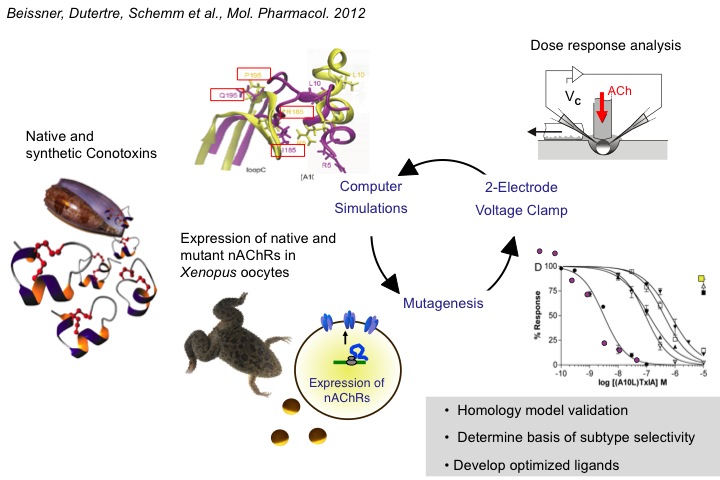
3) Structure-Activity Analysis of a-Conotoxins
In a further project, we study structure-function relationships of a-conotoxins, small peptides from the venom of marine snails. This work aims at developing optimized selective peptide ligands as pharmacological tools for the subtype differentiation of nAChRs. So far, we isolated various novel peptides and determined relations between their sequence and post-translational modifications, and their activity and selectivity (e.g. Nicke et al. J. Biol. Chem. 2003). By rational mutagenesis based on computational docking studies using a homology model derived from the acetylcholine binding protein (AChBP), we could identify amino acid residues in the a- and b-subunits that contribute to the specific interactions with a-conotoxins (Dutertre et al. EMBO J. 2008; Beissner et al. Mol. Pharmacol. 2012).
Methods
- Protein biochemistry (chemical and disulfide cross-linking, BN-PAGE)
- Molecular biology
- Functional analysis of ion channels (uptake measurements, 2-Electrode voltage-clamp, patch-clamp, Voltage clamp fluorometry)
- Generation and characterization of transgenic mice
Förderung durch


AG Nicke 2019

AG Nicke 2016